The inconsistency in phone charging speeds, and the lengthy charging times for electric vehicles, hinges on the movement of lithium ions within a battery’s electrodes. For years, scientists believed they understood this process, but their calculations consistently fell short.
MIT researchers now claim to have solved the problem by rethinking the coordinated movement of electrons and ions. This innovation could lead to the development of batteries that are safer, quicker to charge, and last longer.
Rethinking a Familiar Reaction
The fundamental process within a lithium-ion battery can be visualized as a delicate balancing act. Lithium ions journey through a liquid medium called the electrolyte and embed themselves within solid electrodes, a process termed intercalation. Charging the battery reverses this movement, with the ions flowing in the opposite direction. This cycle, repeated countless times throughout the battery’s life, dictates both the power output and the charging speed.
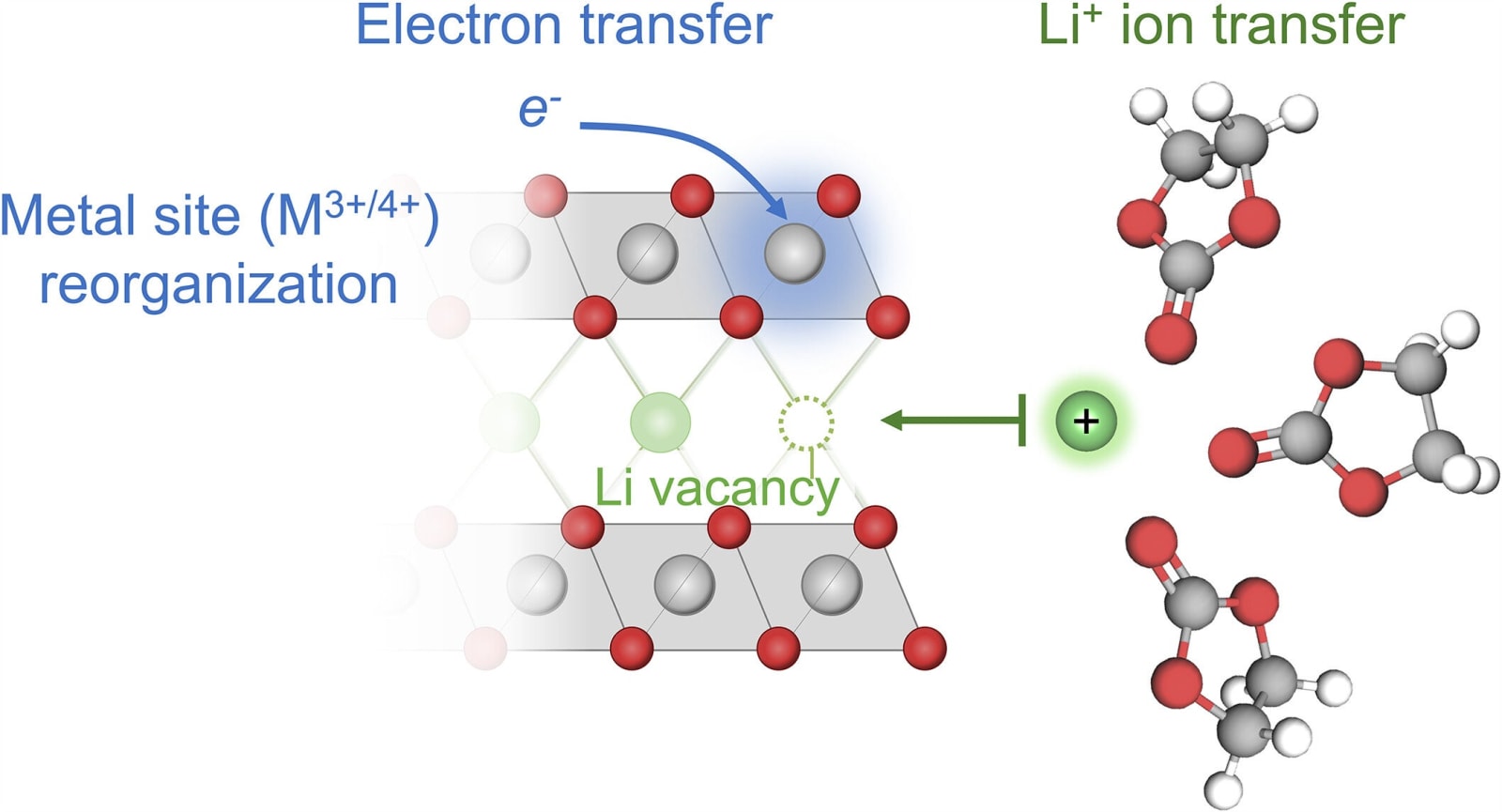
For many years, the Butler-Volmer equation served as the go-to explanation for this reaction, but that’s changed recently. It proposed that the speed of lithium insertion was mainly determined by how quickly ions could travel from the electrolyte into the solid.
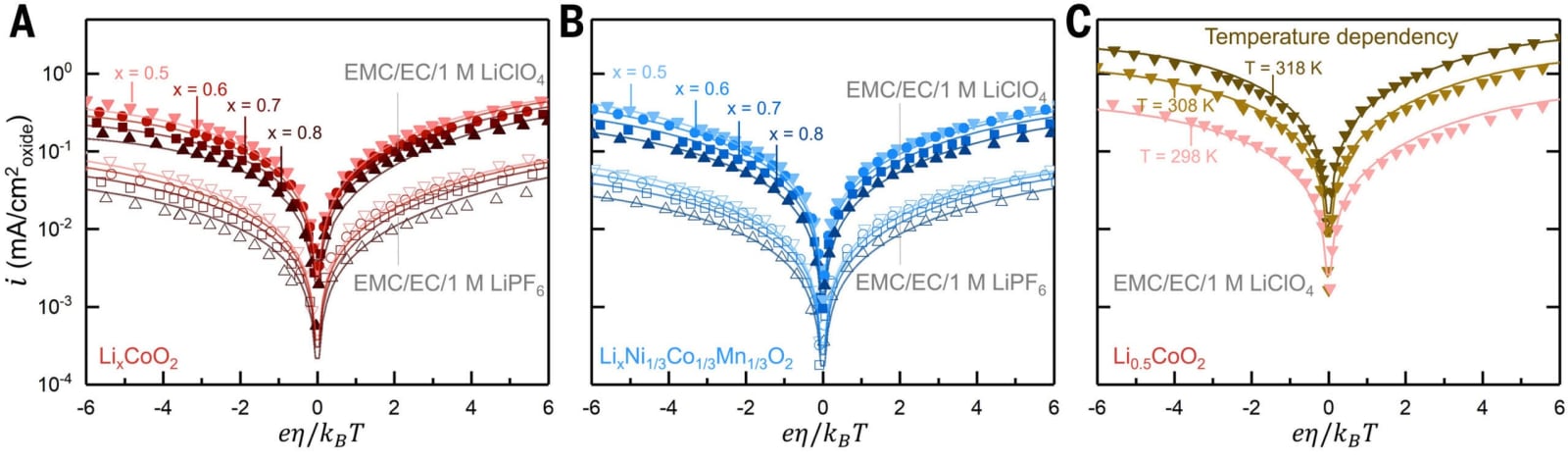
However, things didn’t go as planned. Various labs measuring the same reaction obtained rates that differed by up to a billion times, implying that the conventional model was missing a crucial element.
Testing Over 50 Combinations
Begging questions, the MIT scientistsA sophisticated experiment was designed, where brief voltage bursts were applied to battery electrodes – more like a gentle tap than a hard strike – to observe the behavior of lithium ions and electrons. The researchers didn’t limit themselves to a couple of materials; they explored over 50 different electrode-electrolyte pairings, including commonly used compounds in electric vehicles and portable devices.
The discovery was quite surprising. Intercalation speeds were significantly slower compared to previous reports, and the data didn’t align with Butler-Volmer equation forecasts. This discrepancy led to the development of a novel theoretical framework.
A Two-Part Process: Ion-Electron Transfer in Tandem
The researchers suggested that the incorporation of lithium isn’t simply about ions moving into a solid material. Rather, it’s an interconnected process where an ion can only enter if an electron accompanies it. This synchronized occurrence, termed coupled ion-electron transfer (CIET), requires the simultaneous presence of both species.
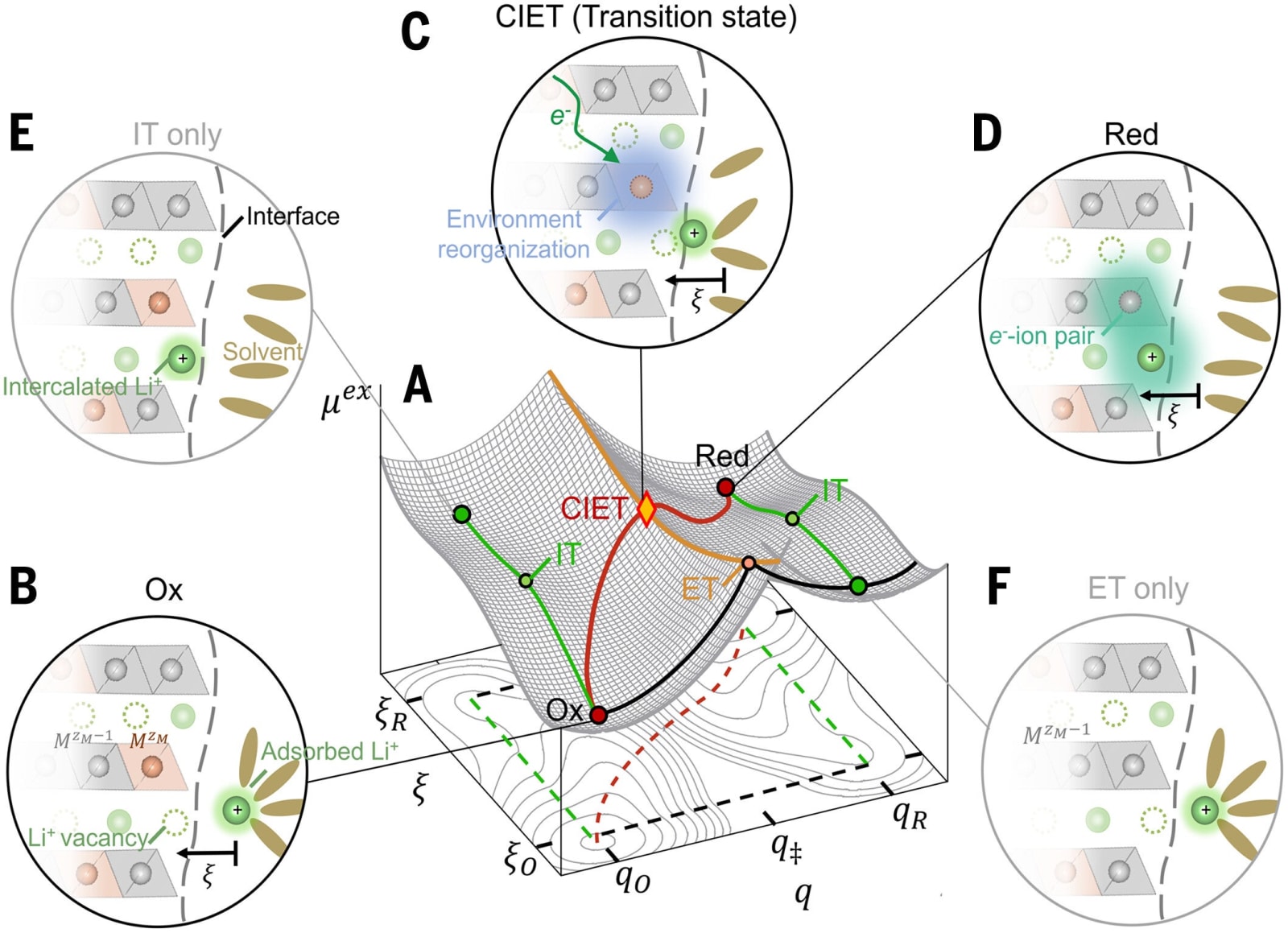
According to MIT’s Martin Bazant, a professor of both chemical engineering and mathematics, “Lithium is intercalated at the same time that the electron is transferred, and they facilitate one another.” By uniting these forces, the reaction’s usual speed bumps are reduced.
The researchers found an almost flawless match between their model’s forecasts and the data from their experiments. Unlike the previous formula, CIET accurately explained the discrepancies observed in measurements across different laboratories.
Fresh Approaches to Battery Construction
Grasping this synergy is incredibly important. If ions and electrons must interact, chemists can now create.electrolytesthat facilitate the combination. Modifying the types of anions present can decrease the energy requirement for the linked movement, allowing batteries to charge more quickly and store more energy.
Yang Shao-Horn, a professor of mechanical engineering, chemistry, and materials science at MIT, identified the potential: “Tuning the intercalation kinetics by changing electrolytes offers excellent opportunities to optimize reaction rates, change electrode architectures, and hence enhance the battery power and energy.”
This understanding also lays the groundwork for minimizing harmful side effects that degrade battery performance over time. While stray electrons leaking into the electrolyte are just one aspect of the problem, now that researchers have a better grasp of transfer processes, engineers can determine how to prevent these losses.
Beyond Trial and Error
To date, most advances in battery technology have arisen from trial and error experimentation. Researchers would try out new materials and see if there was an enhancement in performance or not. With the CIET framework, research can significantly more targeted.
The MIT researchers already automate experiments to test thousands of electrolyte recipes quickly, comparing the results against machine learning models for predicting which chemistries will perform optimally. Instead of making an educated guess, they can now design theoretically.
Co-author of the study Yirui Zhang, who is now a professor at Rice University, and others Dimitrios Fraggedakis at Princeton University, Tao Gao at the University of Utah, and MIT graduate student Shakul Pathak helped build the model. In addition to Bazant and Shao-Horn, they published the findings in Science.
Moving Closer to Quicker Charging Speeds
With a more solid scientific foundation, battery technology could advance. Faster charging mobile phones and laptops, electric cars that can be charged in minutes, not hours, and products that run longer all move from fantasy to possibility.
Challenges remain, since batteries employed in the real world are bedeviled by such as material imperfections and rivalrous reactions. But the CIET model gives scientists a powerful tool to attack such issues head-on.
Bazant summarized the promise: “If you want to do that rationally, not by experimenting, you require some kind of theory to have a notion what are the key material parameters that you can tinker with. That’s what this paper tries to provide.”
Practical Implications of the Research
The new model opens up the doors for building batteries that charge much faster without any sacrifice of safety or longevity.
In everyday life, it would translate into phones, computers, and cars that can be operational in minutes instead of hours. It also implies a way of minimizing side reactions that reduce battery life, keeping the devices active longer and wastefulness to a minimum.
For researchers, the CIET model provides a clear roadmap for the creation of new materials without trial and error. That process can speed up innovation, lower expenses, and ultimately make clean energy technology cheaper worldwide.
Research findings are available online in the journal Science.
Related Stories
・Stop-and-go driving may actually be good for EV batteries
・New EV battery tech could power cars for up to 1 million kilometers
・Graphene innovation significantly improves EV battery capacity and safety
Like these kind of feel good stories? Get The Brighter Side of News’ newsletter.




